Parkinson’s disease candidate tozadenant (A2a antagonist) is a key component of Biotie’s investment case and detailed Phase IIb data indicate a highly competitive profile for the drug. Tozadenant is now Biotie’s primary operational focus, supported by trial milestones from partner UCB, and Phase III studies to start in H115. With €45m in cash and milestones / royalties to flow in from Lundbeck’s EU sales (launched this week) of alcohol-dependence drug Selincro, Biotie’s portfolio review may bring in partners and/or new product opportunities to make best use of these resources. This could add upside to our €231m, or €0.51/share, valuation.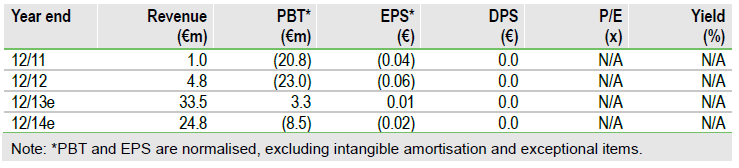
Tozadenant Data Impress
Detailed data presented at AAN last month indicate a highly competitive profile for drug, compared to other A2a antagonists and alternative Parkinson’s disease candidates. Significant reductions in ‘off’ time, increases in ‘on’ time, reductions in UPDRS scores, improvements in clinician/patient assessments and an acceptable safety profile indicate a more robust profile than preladenant and istradefylline.
Backfilling Underway
Biotie has full responsibility for the Phase III programme, funded by UCB payments of over $100m over the next six years; this is additional to the $340m potential clinical/regulatory/commercial milestones payable by UCB to Biotie. The studies will start in H115, while Biotie completes the required manufacturing, toxicology and regulatory work on tozadenant (drawing on UCB’s facilities and expertise).
Selincro Offers A Paradigm Shift
Lundbeck launched Selincro (nalmefene) in Norway, Finland, Poland and the Baltic countries this week, a breakthrough event as Biotie’s first marketed product (further EU launches in 2013-14). Taken as-needed, Selincro reduces alcohol consumption, whereas existing drug therapies target complete abstinence from drinking, which is often not an acceptable or attainable treatment goal. Initial uptake may be slow but the potential is significant given 16m alcoholics across the EU (peak sales €320m).
Valuation: €231m On Increased Tozadenant Probability
We value Biotie at €231m, or €0.51 per share, after raising tozadenant’s probability of success to a Phase III standard of 65% on the strength of the Phase IIb data. Our valuation is based on a risk-adjusted NPV of Biotie’s key products – Selincro, tozadenant and SYN120 – and €45m estimated gross cash at Q113. We see upside potential should Biotie secure a partner(s) for SYN120 (5HT6 antagonist) and/or BTT-1023 (VAP-1 antibody). The ongoing portfolio review, to conclude mid-2013, may bring in fresh product opportunities to build the investment case further.
To Read the Entire Report Please Click on the pdf File Below.
- English (UK)
- English (India)
- English (Canada)
- English (Australia)
- English (South Africa)
- English (Philippines)
- English (Nigeria)
- Deutsch
- Español (España)
- Español (México)
- Français
- Italiano
- Nederlands
- Português (Portugal)
- Polski
- Português (Brasil)
- Русский
- Türkçe
- العربية
- Ελληνικά
- Svenska
- Suomi
- עברית
- 日本語
- 한국어
- 简体中文
- 繁體中文
- Bahasa Indonesia
- Bahasa Melayu
- ไทย
- Tiếng Việt
- हिंदी
Biotie Therapies
Latest comments
Loading next article…
Install Our App
Risk Disclosure: Trading in financial instruments and/or cryptocurrencies involves high risks including the risk of losing some, or all, of your investment amount, and may not be suitable for all investors. Prices of cryptocurrencies are extremely volatile and may be affected by external factors such as financial, regulatory or political events. Trading on margin increases the financial risks.
Before deciding to trade in financial instrument or cryptocurrencies you should be fully informed of the risks and costs associated with trading the financial markets, carefully consider your investment objectives, level of experience, and risk appetite, and seek professional advice where needed.
Fusion Media would like to remind you that the data contained in this website is not necessarily real-time nor accurate. The data and prices on the website are not necessarily provided by any market or exchange, but may be provided by market makers, and so prices may not be accurate and may differ from the actual price at any given market, meaning prices are indicative and not appropriate for trading purposes. Fusion Media and any provider of the data contained in this website will not accept liability for any loss or damage as a result of your trading, or your reliance on the information contained within this website.
It is prohibited to use, store, reproduce, display, modify, transmit or distribute the data contained in this website without the explicit prior written permission of Fusion Media and/or the data provider. All intellectual property rights are reserved by the providers and/or the exchange providing the data contained in this website.
Fusion Media may be compensated by the advertisers that appear on the website, based on your interaction with the advertisements or advertisers.
Before deciding to trade in financial instrument or cryptocurrencies you should be fully informed of the risks and costs associated with trading the financial markets, carefully consider your investment objectives, level of experience, and risk appetite, and seek professional advice where needed.
Fusion Media would like to remind you that the data contained in this website is not necessarily real-time nor accurate. The data and prices on the website are not necessarily provided by any market or exchange, but may be provided by market makers, and so prices may not be accurate and may differ from the actual price at any given market, meaning prices are indicative and not appropriate for trading purposes. Fusion Media and any provider of the data contained in this website will not accept liability for any loss or damage as a result of your trading, or your reliance on the information contained within this website.
It is prohibited to use, store, reproduce, display, modify, transmit or distribute the data contained in this website without the explicit prior written permission of Fusion Media and/or the data provider. All intellectual property rights are reserved by the providers and/or the exchange providing the data contained in this website.
Fusion Media may be compensated by the advertisers that appear on the website, based on your interaction with the advertisements or advertisers.
© 2007-2024 - Fusion Media Limited. All Rights Reserved.