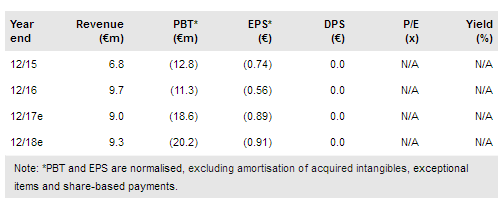
Medigene NA O.N. (DE:MDG1k) has announced that it has submitted a clinical trial authorisation application (CTA) to begin its first clinical trial with its proprietary T-cell receptor (TCR) modified T cells. If approved, the trial will evaluate it as an immunotherapy to treat a range of blood cancer indications and will be one of the first in Germany of TCR-modified cells. The trial is expected to start by the end of 2017. This submission triggers a final milestone payment of €2m for the Trianta acquisition (January 2014). We maintain our valuation of €315m but note the company is well-funded to deliver important milestones from progressing its clinical programme.
Clinical trial application submitted for its TCR CIT
In 2017 we expect Medigene to take significant steps with its TCR programme, primarily with the initiation of its own TCR clinical study. The announcement on 10 July indicates that the company’s plans are on track and, subject to regulatory approval, a combined Phase I/II safety and feasibility clinical trial will start in H217. This is a significant milestone as the company’s first clinical study for its proprietary modified T cells and one of the first TCR trials in Germany. The TCR therapy targets PRAME (preferentially expressed antigen in melanoma) and will be evaluated in patients with advanced haematological diseases. For an overview of the company-initiated trial (CIT) design and Medigene’s TCR technology please see our recent note here.
To read the entire report Please click on the pdf File Below: